 A great episode from the Gravy podcast on how my home state developed a love of cheese dip.
A great episode from the Gravy podcast on how my home state developed a love of cheese dip.
There’s a dish you’ll find at every kind of restaurant in Little Rock, from the pizza places to the burger joints: cheese dip. How did it become so beloved in Arkansas? And what does it reveal about the state’s past—and present? In this episode of Gravy, Dana Bialek and I investigate this story of highways, demographic changes, and a food’s shifting identity over time.
Also here is a convenient map of must visit, cheese dip spots in Arkansas. I’ve visited more of these than I care to admit. Of course a major ingredient of many Arkansas, cheese-dip recipes is a big, oily, orange block of Velveeta.
All this brings me to the age old question of “Is Velveeta one molecule away from plastic?” You have likely heard the same “logic” applied to margarine. Of course, this is wrong in many ways.
Both Velveeta and margarine are composed of many different molecules so such a statement is already meaningless. The ingredient list of Velveeta is milk, whey, skim milk, milk protein concentrate, water, milkfat, whey protein concentrate, sodium phosphate, modified food starch; contains less than 2% of: salt, calcium phosphate, dried corn syrup, canola oil, malto dextrin, lactic acid, sorbic acid as a preservative, sodium alginate, sodium citrate, cheese culture, enzymes, apocarotenal (color), annatto (color). Milk itself, just one of the ingredients listed, contains 12 different fatty acid molecules.
But adding a single molecule to anything could make a big difference. Take for example the difference between hydrogen peroxide H2O2 and water H20, which have only one atom, hydrogen, different.
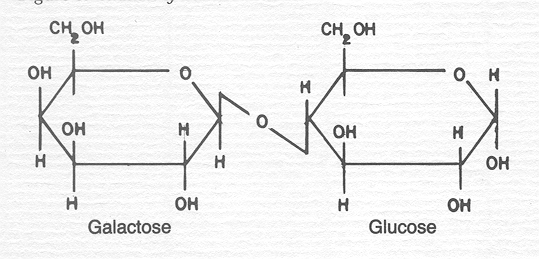 But also the one molecule away is odd because it can take much less to get a significant change. The structure of the molecule can make a big difference without even changing the chemical composition of molecule. Lactose is made out of two simpler sugars, glucose and galactose. The chemical formula for glucose is C6H12O6. The chemical formula for galactose is also C6H12O6. Yes, they are chemically identical. They are structured differently and that gives them different properties.
But also the one molecule away is odd because it can take much less to get a significant change. The structure of the molecule can make a big difference without even changing the chemical composition of molecule. Lactose is made out of two simpler sugars, glucose and galactose. The chemical formula for glucose is C6H12O6. The chemical formula for galactose is also C6H12O6. Yes, they are chemically identical. They are structured differently and that gives them different properties.
